Full HTML
Case Report: Inflammatory Cerebral Amyloid Angiopathy - Recognizing a Treatable Form of Cerebral Vasculitis - Volume 2 Issue 2 (July-Dec), - (6 Months )
Pages: 16-21
Category: Short Report
Published Date: 12-12-2025
Garrett Harris1, Chimaobi Ochulo2, and Arjun Athreya1
Author Affiliation:
1 Division of Neurology at Lexington Medical Center, USA
2 American University of the Caribbean-School of Medicine, USA
∗ Correspondence: Garrett Harris, Email: garrettEharris@gmail.com
Keywords:
Inflammatory Cerebral Amyloid Angiopathy, Cerebral Vasculitis, Boston Criteria 2.0, Neuroimmunology, Cognitive Decline, Immunosuppressive Therapy
Full Text:
1. Introduction
Cerebral amyloid angiopathy (CAA) is an age-associated small vessel disease characterized by gradual deposition of beta-amyloid within the walls of cortical and leptomeningeal vessels. This accumulation leads to a spectrum of clinical manifestations, including spontaneous intracerebral hemorrhage (ICH), transient focal neurological episodes, and cognitive decline [1,2]. However, due to its variable clinical and radiological presentation—and its overlap with other neurovascular and neurodegenerative conditions—CAA is often underdiagnosed, especially in patients who lack typical hemorrhagic signs or present with non-specific neurological symptoms.
Historically considered a post-mortem diagnosis, CAA is now more frequently identified in vivo due to evolving diagnostic criteria. Today, “cerebral amyloid angiopathy” refers not only to a distinctive cerebrovascular pathological process but also includes a constellation of clinical presentations, characteristic lesions on imaging, and diagnostic frameworks such as the Boston Criteria that have enabled recognition in living patients [3]. The diagnostic framework for CAA has evolved substantially with the development and recent revision of the Boston Criteria. The Boston Criteria version 2.0, now adopted as the international standard, incorporates advanced MRI markers—such as strictly lobar cerebral microbleeds, cortical superficial siderosis, severe perivascular spaces, and multispot white matter hyperintensities—to enhance diagnostic sensitivity and specificity across diverse clinical presentations [1,4]. These criteria demonstrate improved accuracy for probable CAA, especially in patients presenting with spontaneous ICH, although performance remains limited in asymptomatic patients or those with isolated cognitive symptoms [5].
Sporadic CAA is the most common form, although many subtypes exist. It encompasses a clinical continuum ranging from asymptomatic individuals with incidental imaging findings to those presenting with life-threatening hemorrhage [6]. Notably, inflammatory cerebral amyloid angiopathy (iCAA) produces subacute encephalopathy, seizures, and headaches with asymmetric white matter edema, and notably responds dramatically to immunosuppressive therapy, particularly corticosteroids [6].
While CAA is predominantly age-related, it can be identified post-mortem by amyloid-beta replacing the walls of small cortical vessels. If pathological changes could be directly visualized during life, diagnosis and prognostication would be more straightforward. Thus, improving in vivo diagnostic tools remains essential for early identification, risk stratification, and therapeutic decision-making [5].
The following case describes an 84-year-old Caucasian woman who presented with progressive aphasia and cognitive decline and who ultimately fulfilled the Boston criteria for probable cerebral amyloid angiopathy. This case is significant in that it highlights the diagnostic complexity of CAA and its complications, particularly when initial presentations are atypical or when there is a delay in recognition.
2. Case Presentation
An 84-year-old right-handed white female with a past medical history significant for coronary artery disease (status post-MI with stent placement), hypertension, hyperlipidemia, hypothyroidism, osteoporosis, depression, anxiety, and irritable bowel syndrome presented to the emergency department with a two-week history of word-finding difficulty and mild receptive aphasia. The patient denied motor symptoms, visual disturbances, or systemic complaints, although she described discomfort as “pain in the back of her eyes.” On initial neurological examination, she was alert and oriented but exhibited expressive aphasia and mild word-finding difficulty. A non-contrast head CT revealed multifocal white matter changes and concern for vasogenic edema with possible underlying mass lesions. Brain MRI demonstrated extensive bilateral subcortical and periventricular T2/FLAIR hyperintensities with relative cortical sparing, along with a non-enhancing 1.3 cm lesion in the right parietal lobe that contains proteinaceous and hemorrhagic material. Additionally, widespread cortical microhemorrhages were noted. Overall findings were believed to represent a combination of chronic microvascular disease and isolated hemorrhage.
Neurosurgical consultation determined the lesion to be most consistent with a small parietal intracerebral hemorrhage (ICH), likely related to cerebral amyloid angiopathy (CAA), particularly given gradient echo findings suggestive of microhemorrhages as previously stated. No surgical intervention was recommended. She was initially treated with IV dexamethasone for vasogenic edema, which was discontinued prior to discharge, and told to follow up with neurology outpatient, which she did in 2020 during the COVID-19 pandemic. However, after several years, the patient returned to the emergency department with increased confusion and acute encephalopathy. According to her daughter, she had become increasingly disoriented, demonstrating repetitive speech and difficulty with activities of daily living. Neurological examination revealed fluctuating attention, marked expressive aphasia, and impaired comprehension. There were no motor deficits, no new visual complaints, and no evidence of seizure activity. A repeat brain MRI revealed more extensive T2 FLAIR white matter changes in different distributions than prior, raising suspicion for an inflammatory process and/or vasogenic edema. The cortical microhemorrhages were also re-demonstrated. Given the progressive clinical deterioration and radiographic evolution, a broader differential was revisited. Initially, infectious etiologies, progressive multifocal leukoencephalopathy (PML), and low-grade neoplasm were considered but were systematically excluded through a workup that included serologies and cerebrospinal fluid analysis. In the absence of an alternate etiology, the presence of T2 FLAIR white matter abnormalities in conjunction with widespread cortical microhemorrhages raised the possibility of an iCAA variant.
Given this diagnostic uncertainty, neurosurgery was consulted. A brain biopsy was discussed but ultimately deferred in favor of empiric immunosuppressive therapy. The patient was initiated on intravenous methylprednisolone 500 mg twice daily for five days, followed by a prednisone taper. Two months after initiating immunosuppressive therapy, a follow-up MRI revealed reduced vasogenic edema and white matter changes with stable microhemorrhages. These findings reinforced the diagnosis of iCAA without the need for supporting pathology confirmation.
At a subsequent follow-up two months later, her family and clinical reports described improvements in communication and daily functioning. A follow-up MRI confirmed continued resolution of vasogenic edema and absence of new hemorrhagic lesions. In the months that followed, neurology concluded that the patient had returned to her baseline cognitive function. The family denied new memory loss, and the neurological exam was non-focal, with the absence of aphasia, seizures, or behavioral changes. Mild functional improvement was noted, apart from expected age-related cognitive decline.
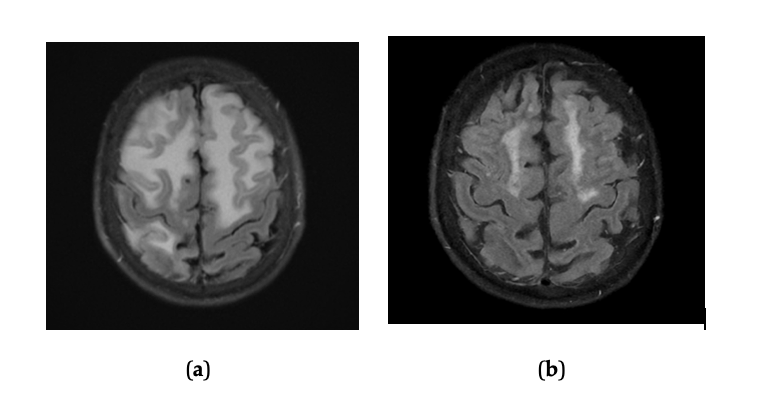
Figure 1. T2 FLAIR MRI of the temporo-occipital lobe showing microbleeds pre-treatment prior and post treatment. (a) T2 FLAIR Sequence of a frontal and parietal lobe section, showing worsening hyperintensities prior to treatment with corticosteroids. (b) T2 FLAIR Sequence of a frontal and parietal lobe section of the brain showing reduction in hyperintensities after treatment with corticosteroids.
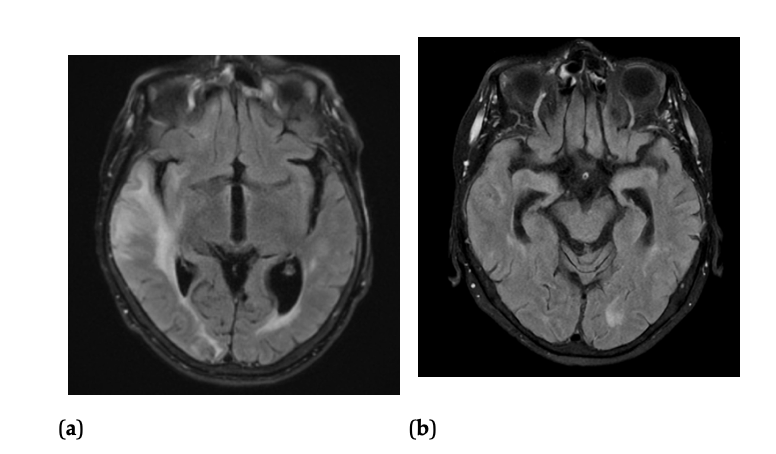 Figure 2. T2 FLAIR MRI of the frontal and parietal lobe showing microbleeds pre-treatment and post treatment (a) T2 FLAIR Sequence of a temporo-occipital lobe section of the brain, showing worsening hyperintensities prior to treatment with corticosteroids. (b) T2 FLAIR Sequence of a temporo-occipital lobe section of the brain, showing reduction in hyperintensities after treatment with corticosteroids.
Figure 2. T2 FLAIR MRI of the frontal and parietal lobe showing microbleeds pre-treatment and post treatment (a) T2 FLAIR Sequence of a temporo-occipital lobe section of the brain, showing worsening hyperintensities prior to treatment with corticosteroids. (b) T2 FLAIR Sequence of a temporo-occipital lobe section of the brain, showing reduction in hyperintensities after treatment with corticosteroids.
3. Discussion
iCAA is a rare condition that is often sporadic, as opposed to isolated CAA, which can be seen in many patients with Alzheimer’s disease and ICH. It may lead to chronic neurological sequelae if not promptly diagnosed and treated. iCAA often presents acutely or subacutely with encephalopathy, focal deficits, and seizures [7–9]. The clinical and radiologic overlap between iCAA and unrelated conditions such as PML, neoplasm, and CNS infections creates a diagnostic challenge in older adults with comorbidities.
Neuroimaging is critical in differentiating iCAA. It is characterized by patchy T2/FLAIR white matter hyperintensities, lobar microbleeds, and cortical superficial siderosis [10,11]. Initially, our patient’s findings were attributed to chronic microvascular disease rather than superimposed inflammation. However, progressive vasogenic edema in the absence of neoplasm or infection raised suspicion for iCAA. A baseline diagnosis of CAA was further supported by Boston Criteria 2.0.
Early recognition of iCAA is essential, as it is a treatable cause of rapidly progressive cognitive decline. The patient improved clinically and radiographically following high-dose corticosteroids and taper. This outcome is consistent with several studies, including a case series of 48 iCAA patients showing 94% clinical improvement and 86% radiographic improvement following immunosuppression [12]. An observational study of 113 patients further reported 88% clinical improvement within three months and lower relapse rates in patients receiving tapering therapy [11,13].
The 6-year delay in this patient’s diagnosis was due to follow-up loss and attribution of symptoms to isolated ICH. Despite these challenges, this case emphasizes the need to consider iCAA in older adults with progressive cognitive decline and evolving white matter changes on imaging.
4. Conclusion
Inflammatory cerebral amyloid angiopathy (iCAA) remains an underrecognized but clinically significant cause of rapidly progressive cognitive decline in older adults. This case shows the diagnostic challenges iCAA poses, especially when initial imaging findings are misattributed to chronic microvascular disease or isolated intracerebral hemorrhage. In patients over 50 with evolving encephalopathy, progressive white matter changes, and lobar microbleeds, iCAA should be considered early, especially when ruling out the more common etiologies. Serial MRI findings, particularly on T2/FLAIR and GRE sequences, can emphasize subtle transitions from expected non-inflammatory CAA to iCAA, guiding more targeted evaluation. In this case, evolving radiographic features prompted a shift in clinical thinking that ultimately allowed for the correct therapeutic intervention. iCAA offers a window for recovery if recognized in time. The patient’s marked improvement following immunosuppressive therapy reinforces this point. Clinicians should note this case as a reminder of the importance of recognizing when a patient’s clinical course diverges from expectations and re-evaluating prior diagnostic assumptions to uncover potentially reversible neurological diseases. In iCAA, timely recognition and early diagnosis opens the door to effective treatment and functional improvement.
References:
Charidimou A, Boulouis G, Frosch MP, et al. The Boston criteria version 2.0 for cerebral amyloid angiopathy: a multicentre, retrospective, MRI–neuropathology diagnostic accuracy study. Lancet Neurology. 2022;21(8):714–725.
Kozberg MG, Perosa V, Gurol ME, van Veluw SJ. A practical approach to the management of cerebral amyloid angiopathy. International Journal of Stroke. 2021;16(4):356–369.
Banerjee G, Carare R, Cordonnier C, et al. The increasing impact of cerebral amyloid angiopathy: essential new insights for clinical practice. Journal of Neurology, Neurosurgery & Psychiatry. 2017;88(11):982–994.
Charidimou A, Boulouis G, Gurol ME, et al. Emerging concepts in sporadic cerebral amyloid angiopathy. Brain. 2017;140(7):1829–1850.
Greenberg SM, Charidimou A. Diagnosis of cerebral amyloid angiopathy: evolution of the Boston Criteria. Stroke. 2018;49(2):491–497.
Vinters HV. Cerebral amyloid angiopathy: a critical review. Stroke. 1987;18(2):311–324.
Chung Kong Khi, Anderson NE, Hutchinson D, et al. Cerebral amyloid angiopathy–related inflammation: three case reports and a review. Journal of Neurology, Neurosurgery & Psychiatry. 2011;82(1):20–26.
Salvarani C, Hunder GG, Morris JM, Brown RD Jr, Christianson T, Giannini C. Aβ-related angiitis: comparison with cerebral amyloid angiopathy without inflammation and primary CNS vasculitis. Neurology. 2013;81:1596–1603.
Jack CR Jr, Andrews JS, Beach TG, et al. Revised criteria for diagnosis and staging of Alzheimer’s disease: Alzheimer’s Association Workgroup. Alzheimer’s & Dementia. 2024;20(8):5143–5169.
de Souza A, Tasker K. Inflammatory cerebral amyloid angiopathy: a broad clinical spectrum. Journal of Clinical Neurology. 2023;19(3):230–241.
Theodorou A, Palaiodimou L, Malhotra K, et al. Clinical, neuroimaging, and genetic markers in cerebral amyloid angiopathy–related inflammation: a systematic review and meta-analysis. Stroke. 2023;54(1):178–188.
Todd R, McKenna E, McCluskey G, et al. Epidemiologic, clinical, and radiologic study of cerebral amyloid angiopathy–related inflammation in Northern Ireland. Neurology. 2025;105(5):e214005.
Downes MH, Kalagara R, Rossitto CP, et al. Validation of the Boston Criteria Version 2.0 for cerebral amyloid angiopathy in patients presenting with intracerebral hemorrhage. Neurology. 2025;104(7):e213460.