Full HTML
Diagnostic Accuracy of Meningitis/Encephalitis (ME) Panel for Central Nervous System Infections: An Updated Meta-Analysis - Volume 2 Issue 2 (July-Dec), - (6 Months )
Pages: 24-38
Category: Original Research
Published Date: 30-12-2025
Ralph Louie Bautista
Author Affiliation:
1 Department of Physiology, College of Medicine, University of the East-Ramon Magsaysay Memorial Medical Center Inc., Aurora Boulevard, Quezon City 1113, Philippines
2 Department of Neurosciences, Department of Health East Avenue Medical Center, Diliman, Quezon City, Philippine
Keywords:
CNS infections, meningitis, encephalitis, multiplex PCR, diagnostic accuracy, ME panel, FilmArray, meta-analysis
Full Text:
Introduction
Central nervous system infections (CNSI), including meningitis and encephalitis, represent significant causes of morbidity and mortality worldwide[1,2,3]. Often, the early clinical symptoms are nonspecific, which pose a diagnostic challenge for clinicians especially in resource-limited settings. To prevent severe complications and death from CNSI, timely and accurate detection and characterization of pathogens through laboratory testing are crucial.
In recent years, diagnostics in infectious diseases have transitioned from conventional methods to novel technologies that utilize molecular assays capable of identifying presence of pathogens within hours[4]. The first commercial multiplex polymerase chain reaction (PCR) tool for microbial detection---known as the BioFire® FilmArray® Meningitis/Encephalitis (ME) Panel (BioFire Diagnostics, Salt Lake City, Utah)---was introduced in 2015, which allowed for the simultaneous detection of 14 pathogens from CSF samples. These include 6 bacteria (Streptococcus pneumoniae, Neisseria meningitidis, Haemophilus influenzae, Listeria monocytogenes, Streptococcus agalactiae, Escherichia coli), 7 viruses (Herpes simplex virus 1 [HSV1], Herpes simplex virus 2 [HSV2], Varicella Zoster virus [VZV], Cytomegalovirus [CMV], Human herpesvirus 6 [HHV6], Human Parechovirus [HPeV], and Enterovirus), and 1 yeast (Cryptococcus neoformans/gattii). Studies have demonstrated the ME Panel’s ability to reduce time to diagnosis (TTD) and comparable detection of CNS pathogens with traditional methods such as cultures that have slow turnaround times. Various studies have assessed the diagnostic accuracy of the ME panel against culture studies, showing an overall agreement rate ranging from 93% to 99% along with high sensitivity and specificity[5]. Moreover, the ME panel only requires approximately 200 microliter (uL) of CSF specimen[6], thereby potentially avoiding the challenges of multiple assays. Over the years, the ME panel has been increasingly used worldwide due to its clinical utility in diagnosing CNSI.
Previous meta-analyses on commercial ME panels were predominantly drawn from studies conducted in high-income or upper-middle-income countries[7,8]. While the adoption of this technology has expanded globally, data on the clinical utility of the ME panel in low-income and lower-middle-income countries remain scarce[9]. This is particularly concerning given that the burden of CNSI is significantly higher in these regions due to factors such as denser living conditions, lower vaccination coverage, and higher prevalence of co-existing health conditions. The lack of robust data from these healthcare settings presents a critical gap in understanding the full potential and challenges of implementing ME panels where they are most needed. This study aims to expand the evidence base by including studies from a broader range of geographic and economic settings, assessing the ME panel's diagnostic accuracy across diverse healthcare environments. As technological advancements and pathogen dynamics continue to evolve, an updated analysis could provide valuable insights into optimizing the use of ME panels for CNSI in a wider array of healthcare contexts.
Given the importance of accurate and timely diagnosis in the management of CNSI, it is essential to explore and assess the effectiveness of both traditional and modern diagnostic methods. Understanding how these methods perform in different clinical settings, especially where resources are limited, is crucial for improving the delivery of care.
Objectives
The primary objective of this study is to provide an updated and comprehensive analysis of the diagnostic accuracy of the commercial multiplex ME panel for each pathogen across diverse healthcare settings. Additionally, it seeks to fill gaps in existing research by incorporating data from low- and lower-middle-income countries, where the burden of CNS infections is greater, and where the implementation of ME panels may face distinct challenges. This expanded approach will offer a more complete understanding of the ME panel’s performance in both resource-rich and resource-limited environments.
Materials and Methods
Protocol
A detailed presentation and description of selected articles used in this review were outlined in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement.
Eligibility Criteria
The researcher applied specific inclusion criteria to ensure the relevance and quality of the selected studies. Articles were included if they met the following conditions: (1) they focused on the utilization of cerebrospinal fluid (CSF) in healthcare settings, such as hospitals, (2) employed molecular methods for the identification of microbial pathogens, (3) involved in vivo or in vitro studies, (4) were full-text publications, and (5) were written in English. Conversely, studies were excluded if they did not take place in healthcare environments, lacked molecular analysis, or were published in languages other than English. No restrictions were imposed on the year of publication, allowing for a comprehensive review of the literature across all time periods. This approach was designed to capture the most relevant and methodologically sound research on the topic.
Information sources, Search Strategy, and Selection Process
In September 2024, a comprehensive electronic search in PubMed, Web of Science, and Embase was performed using keywords such as (CNS infection) (Meningitis/ Encephalitis Panel) (FilmArray) (BioFire) (PCR) OR (culture) AND (CSF) following the PRISMA protocol (Figure 1). The search was restricted to studies investigating CNSI using the BioFire® FilmArray® Meningitis/Encephalitis (ME) Panel. The inclusion criteria encompassed both in vivo and in vitro studies published in English. Non-English papers and studies that did not meet the predefined criteria were excluded. The database search identified 2022 articles potentially applicable for the analysis. After eliminating all duplications, 67 articles were screened. The researcher excluded 44 articles as the articles did not focus on the topic of this review. In total, 23 articles were then subjected to a full-text analysis.
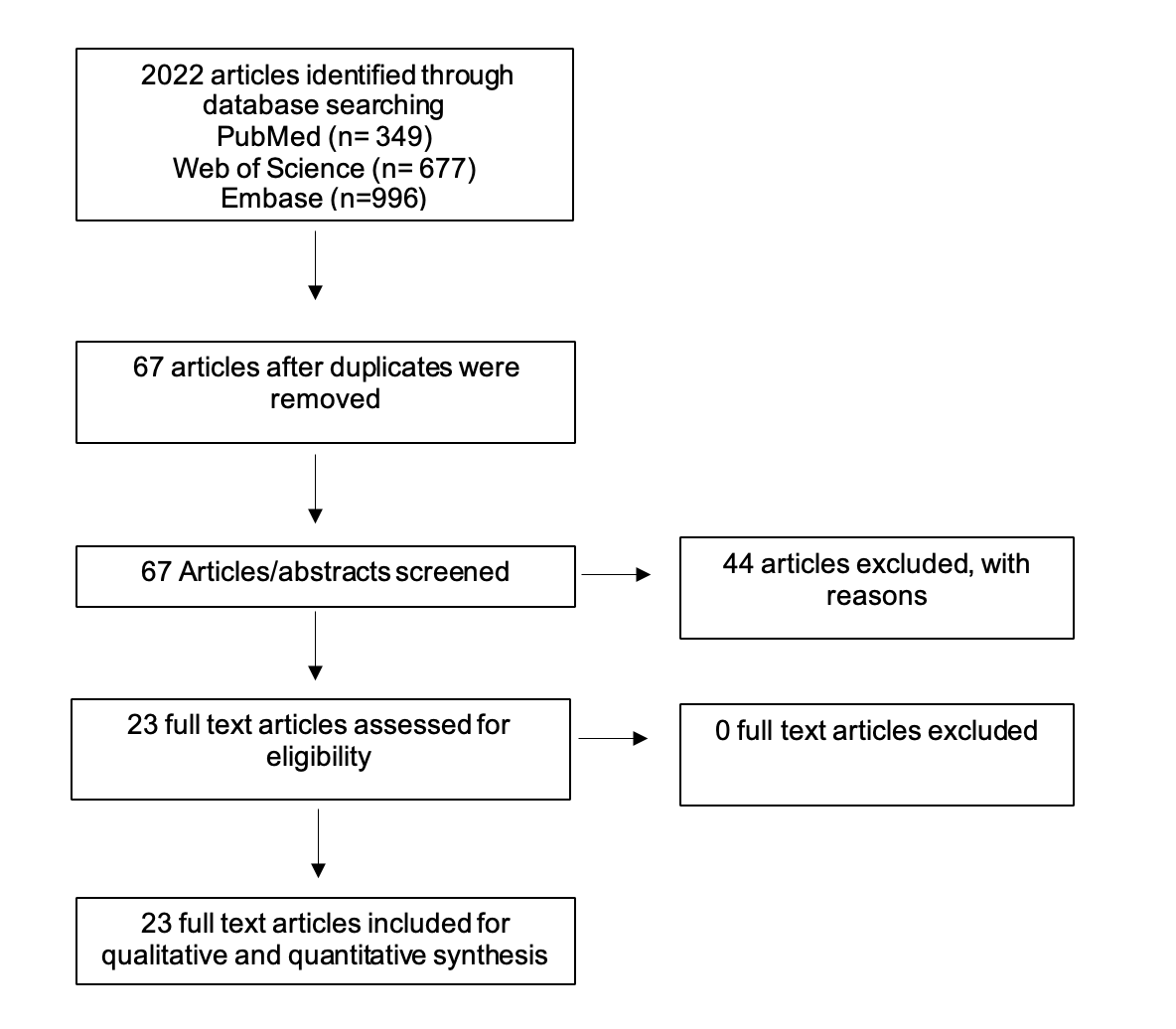
Results
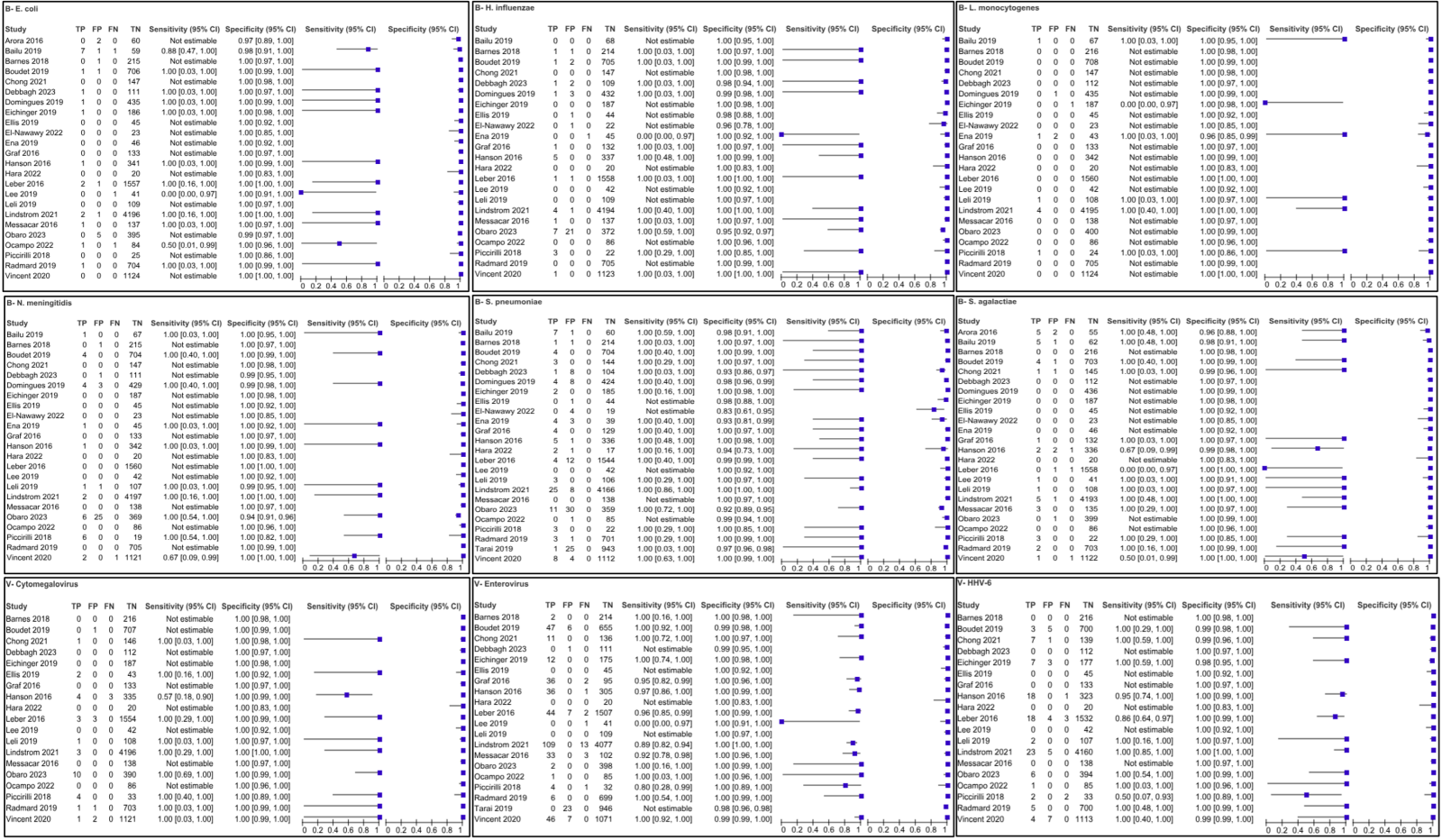
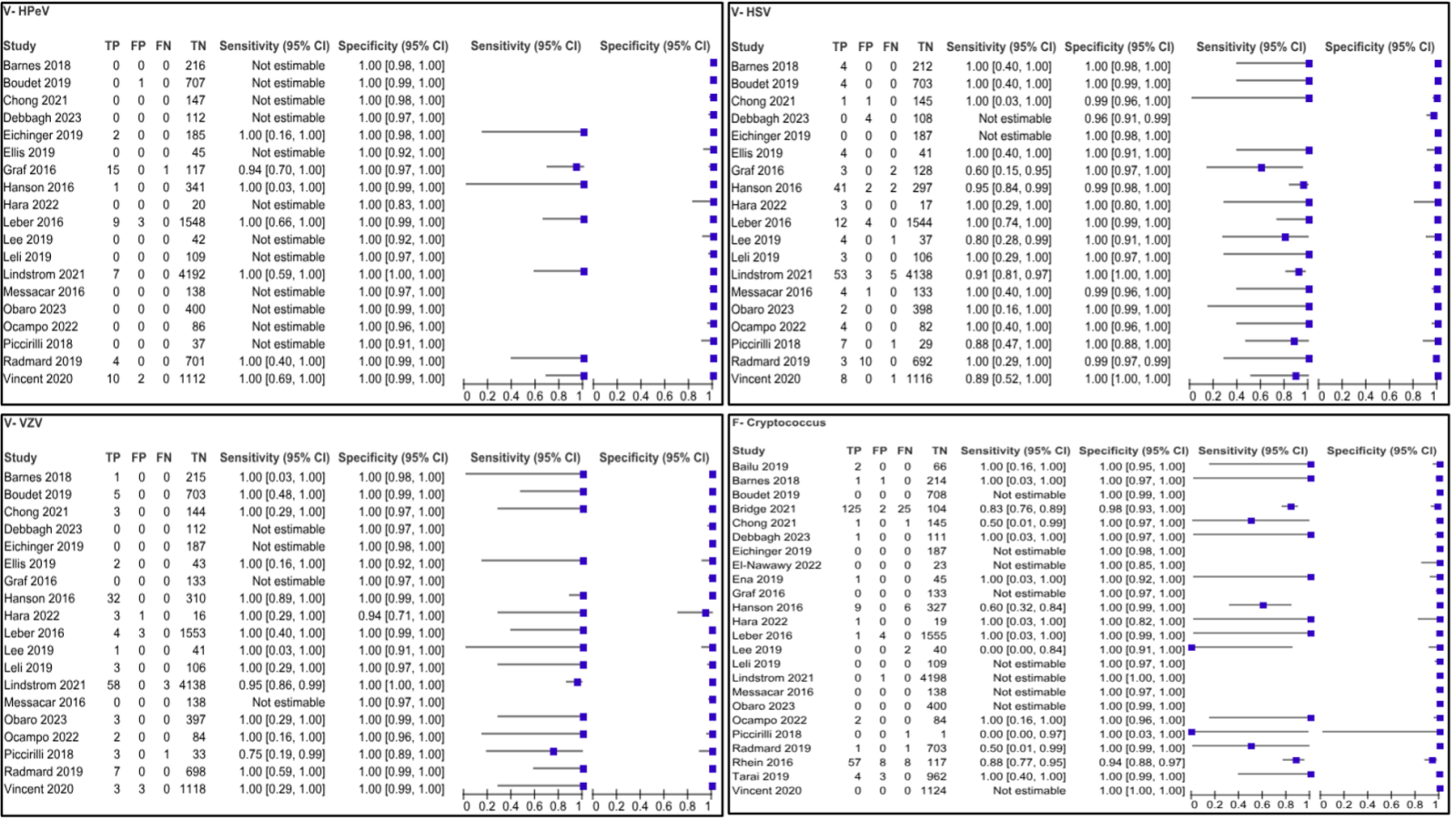
There were a total of 2,022 articles screened, of which 23 were included in the quantitative synthesis (Figure 1). The main characteristics of the included studies are summarized in Table 1. The diagnostic performance of the BioFire ME panel shows notable variability across different pathogens, reflecting both its strengths and limitations in clinical use.
Table 1. Summary of studies included in the meta-analysis (Part 1)
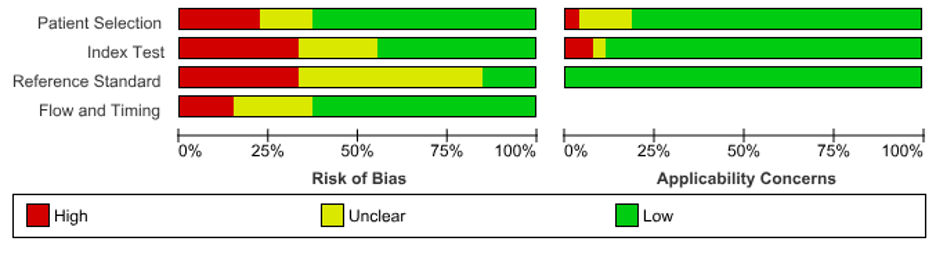
Risk of Bias
Shows the risk of bias and applicability concerns in all studies included. The four key domains of the Quality Assessment of Diagnostic Accuracy Studies (QUADAS-2) was applied [10] (Figure 4).
Discussion
Overall, the ME Panel consistently demonstrated strong diagnostic capabilities, particularly for viral pathogens, with both sensitivity and specificity often exceeding 90%.
Conclusion
The BioFire® FilmArray® ME Panel demonstrates high diagnostic accuracy, particularly for viral pathogens.
Conclusions
This section is not mandatory, but can be added to the manuscript if the discussion is unusually long or complex.
Conflict of Interest
The authors declare no conflict of interest.
References:
- Wunrow HY, Benlder RG, Vongpradith A, Sirota SB, Swetschinski LR, Novotney A, Gray AP, Ikuta KS, Sharara F, Wool EE, Aali A. Global, regional, and national burden of meningitis and its aetiologies, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. The Lancet Neurology. 2023;22(8):685–711. https://doi.org/10.1016/S1474-4422(23)00195-3 [Google Scholar] [PubMed]
- Wang H, Zhao S, Wang S, et al. Global magnitude of encephalitis burden and its evolving pattern over the past 30 years. Journal of Infection. 2022;84(6):777–787. https://doi.org/10.1016/j.jinf.2022.04.026 [Google Scholar] [PubMed]
- Zunt JR, Kassebaum NJ, Blake N, Glennie L, Wright C, Nichols E, Abd-Allah F, Abdela J, Abdelalim A, Adamu AA, Adib MG. Global, regional, and national burden of meningitis, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. The Lancet Neurology. 2018;17(12):1061–1082. https://doi.org/10.1016/S1474-4422(18)30387-9 [Google Scholar] [PubMed]
- Dumkow LE, Worden LJ, Rao SN. Syndromic diagnostic testing: a new way to approach patient care in the treatment of infectious diseases. Journal of Antimicrobial Chemotherapy. 2021;76(Suppl 3):iii2–iii9. https://doi.org/10.1093/jac/dkab247 [Google Scholar] [PubMed]
- Radmard S, Reid S, Ciryam P, Boubour A, Ho N, Zucker J, Sayre D, Greendyke WG, Miko BA, Pereira MR, Whittier S. Clinical utilization of the FilmArray meningitis/encephalitis (ME) multiplex polymerase chain reaction (PCR) assay. Frontiers in Neurology. 2019;10:281. https://doi.org/10.3389/fneur.2019.00281 [Google Scholar] [PubMed]
- Hemmert A, DesJarlais S, McKinley K, Bourzac K, Holmberg K, Crisp R, Schreckenberger P. Comprehensive testing of CSF specimens using the FilmArray ME panel identifies viral infections overlooked using current clinical practices. Presented at ECCMID Conference, 2015. DOI: N/A [Google Scholar] [PubMed]
- Trujillo-Gómez J, Tsokani S, Arango-Ferreira C, Atehortúa-Muñoz S, Jimenez-Villegas MJ, Serrano-Tabares C, Veroniki AA, Florez ID. Biofire FilmArray Meningitis/Encephalitis panel for the aetiological diagnosis of central nervous system infections: a systematic review and diagnostic test accuracy meta-analysis. EClinicalMedicine. 2022;44:101275. https://doi.org/10.1016/j.eclinm.2022.101275 [Google Scholar] [PubMed]
- Tansarli GS, Chapin KC. Diagnostic test accuracy of the BioFire FilmArray meningitis/encephalitis panel: a systematic review and meta-analysis. Clinical Microbiology and Infection. 2020;26(3):281–290. https://doi.org/10.1016/j.cmi.2019.11.016 [Google Scholar] [PubMed]
- Ocampo FF, Laxamana LC. Diagnostic accuracy of the FilmArray Meningitis/Encephalitis panel in adult patients with suspected bacterial meningitis in a tertiary care hospital in the Philippines. Acta Medica Philippina. 2022;56(13). https://doi.org/10.47895/amp.v56i13.4485 [Google Scholar] [PubMed]
- Whiting P, Rutjes A, Marie W, Mallet S, Deeks J, Reitsma J, et al. QUADAS-2: A revised tool for the quality assessment of diagnostic accuracy studies. Annals of Internal Medicine. 2011;155(8):529–536. https://doi.org/10.7326/0003-4819-155-8-201110180-00009 [Google Scholar] [PubMed]
- Bårnes GK, Gudina EK, Berhane M, Abdissa A, Tesfaw G, Abebe G, et al. New molecular tools for meningitis diagnostics in Ethiopia - a necessary step towards improving antimicrobial prescription. BMC Infectious Diseases. 2018;18(1):684. https://doi.org/10.1186/s12879-018-3561-5 [Google Scholar] [PubMed]
- El-Nawawy AA, Antonios MA, Tawfik ME, Meheissen MA. Comparison of a point-of-care FilmArray test to standard-of-care microbiology test in diagnosis of healthcare-associated infections in a tertiary care pediatric intensive care unit. Antibiotics. 2022;11(4):487. https://doi.org/10.3390/antibiotics11040487 [Google Scholar] [PubMed]
- Obaro S, Hassan-Hanga F, Medugu N, Olaosebikan R, Olanipekun G, Jibir B, Gambo S, Ajose T, Duru C, Ebruke B, Davies HD. Comparison of bacterial culture with BioFire FilmArray multiplex PCR screening of archived cerebrospinal fluid specimens from children with suspected bacterial meningitis in Nigeria. BMC Infectious Diseases. 2023;23(1):68. https://doi.org/10.1186/s12879-023-07924-4 [Google Scholar] [PubMed]
- Debbagh F, Harrar S, Babokh F, Hanchi AL, Soraa N. The contribution of multiplex polymerase chain reaction in the diagnosis of central nervous system infections in intensive care units. Cureus. 2023;15(2):e35168. https://doi.org/10.7759/cureus.35168 [Google Scholar] [PubMed]
- Ellis J, Bangdiwala AS, Cresswell FV, Rhein J, Nuwagira E, Ssebambulidde K, Tugume L, Rajasingham R, Bridge SC, Muzoora C, Meya DB. The changing epidemiology of HIV-associated adult meningitis, Uganda 2015–2017. Open Forum Infectious Diseases. 2019;6(10):ofz419. https://doi.org/10.1093/ofid/ofz419 [Google Scholar] [PubMed]
- Arora HS, Asmar BI, Salimnia H, Agarwal P, Chawla S, Abdel-Haq N. Enhanced identification of group B streptococcus and Escherichia coli in young infants with meningitis using the Biofire Filmarray Meningitis/Encephalitis panel. Pediatric Infectious Disease Journal. 2017;36(7):685–687. https://doi.org/10.1097/INF.0000000000001528 [Google Scholar] [PubMed]
- Bailu D, Hua C, Xia Y, Li J, Xie Y, Tao Y, et al. Evaluation of the BioFire FilmArray meningitis/encephalitis panel for the detection of bacteria and yeast in Chinese children. Annals of Translational Medicine. 2019;7(18):437. https://doi.org/10.21037/atm.2019.08.64 [Google Scholar] [PubMed]
- Boudet A, Pantel A, Carles MJ, Boclé H, Charachon S, Enault C, et al. A review of a 13-month period of FilmArray Meningitis/Encephalitis panel implementation as a first-line diagnosis tool at a university hospital. PLOS ONE. 2019;14(10):e0223887. https://doi.org/10.1371/journal.pone.0223887 [Google Scholar] [PubMed]
- Chong BSW, Kennedy KJ. Comparison of a commercial real-time PCR panel to routine laboratory methods for the diagnosis of meningitis-encephalitis. Pathology. 2021;53(5):644–651. https://doi.org/10.1016/j.pathol.2021.03.006 [Google Scholar] [PubMed]
- Domingues RB, Santos MV, Leite FB, Senne C. FilmArray Meningitis/Encephalitis (ME) panel in the diagnosis of bacterial meningitis. Brazilian Journal of Infectious Diseases. 2019;23(6):405–409. https://doi.org/10.1016/j.bjid.2019.10.003 [Google Scholar] [PubMed]
- Eichinger A, Hagen A, Meyer-Bühn M, Huebner J. Clinical benefits of introducing real-time multiplex PCR for cerebrospinal fluid as routine diagnostic at a tertiary care pediatric center. Infection. 2019;47(1):51–58. https://doi.org/10.1007/s15010-018-1216-5 [Google Scholar] [PubMed]
- Hanson KE, Slechta ES, Killpack JA, Heyrend C, Lunt T, Daly JA, et al. Preclinical assessment of a fully automated multiplex PCR panel. Journal of Clinical Microbiology. 2016;54(3):785–787. https://doi.org/10.1128/JCM.02850-15 [Google Scholar] [PubMed]
- Hara M, Ishihara M, Nakajima H. Use of the FilmArray Meningitis/Encephalitis panel to detect pathogenic microorganisms in cerebrospinal fluid specimens: a single-center retrospective study. Journal of International Medical Research. 2022;50(10). https://doi.org/10.1177/03000605221129261 [Google Scholar] [PubMed]
- Leber AL, Everhart K, Balada-Llasat JM, Cullison J, Daly J, Holt S, et al. Multicenter evaluation of BioFire FilmArray Meningitis/Encephalitis Panel. Journal of Clinical Microbiology. 2016;54(9):2251–2261. https://doi.org/10.1128/JCM.00730-16 [Google Scholar] [PubMed]
- Lee SH, Chen SY, Chien JY, Lee TF, Chen JM, Hsueh PR. Usefulness of the FilmArray meningitis/encephalitis panel for diagnosis in Taiwan. Journal of Microbiology, Immunology and Infection. 2019;52(5):760–768. https://doi.org/10.1016/j.jmii.2018.01.004 [Google Scholar] [PubMed]
- Leli C, Gotta F, Vay D, Calcagno L, Callegari T, Cassinari M, et al. Diagnostic accuracy of a commercial multiplex PCR. Infezioni in Medicina. 2019;27(2):171–179. DOI: N/A [Google Scholar] [PubMed]
- Lindström J, Elfving K, Lindh M, Westin J, Studahl M. Assessment of the FilmArray ME panel in 4199 cerebrospinal fluid samples. Clinical Microbiology and Infection. 2022;28(1):121.e1–121.e6. https://doi.org/10.1016/j.cmi.2021.05.030 [Google Scholar] [PubMed]
- Messacar K, Breazeale G, Robinson CC, Dominguez SR. Potential clinical impact of the FilmArray meningitis/encephalitis panel in children. Diagnostic Microbiology and Infectious Disease. 2016;86(1):109–111. https://doi.org/10.1016/j.diagmicrobio.2016.07.005 [Google Scholar] [PubMed]
- Piccirilli G, Chiereghin A, Gabrielli L, Giannella M, Squarzoni D, Turello G, et al. Infectious meningitis/encephalitis evaluation of multiplex PCR. New Microbiologica. 2018;41(2):103–107. DOI: N/A [Google Scholar] [PubMed]
- Radmard S, Reid S, Ciryam P, Boubour A, Ho N, Zucker J, et al. Clinical utilization of the FilmArray meningitis/encephalitis assay. Frontiers in Neurology. 2019;10:281. https://doi.org/10.3389/fneur.2019.00281 [Google Scholar] [PubMed]
- Vincent JJ, Zandotti C, Baron S, Kandil C, Levy PY, Drancourt M, et al. Point-of-care multiplexed diagnosis of meningitis using the FilmArray ME panel technology. European Journal of Clinical Microbiology & Infectious Diseases. 2020;39(8):1573–1580. https://doi.org/10.1007/s10096-020-03859-9 [Google Scholar] [PubMed]